Our Services
We are designed to strengthen regulatory compliance, enhance operational excellence, and protect the integrity of pharmaceutical businesses. Our approach combines deep industry knowledge with practical execution to ensure your operations remain compliant, efficient, and future-ready.
What We Offer
Regulatory Affairs Consulting

Delivering guidance on regulatory affairs consultancy, including product registration, regulatory submissions, GMP compliance support, and lifecycle management to ensure faster approvals and market success.
Audits and Mock Inspections

We conduct comprehensive audits and mock inspections to assess compliance gaps, strengthen GMP adherence, and ensure your organization is fully prepared for regulatory inspections with confidence and precision.
Stakeholder Engagement

Helping clients on stakeholder engagement services that enhance collaboration with regulatory authorities, industry partners, and key decision-makers, and compliance alignment.
Clinical Trial Application
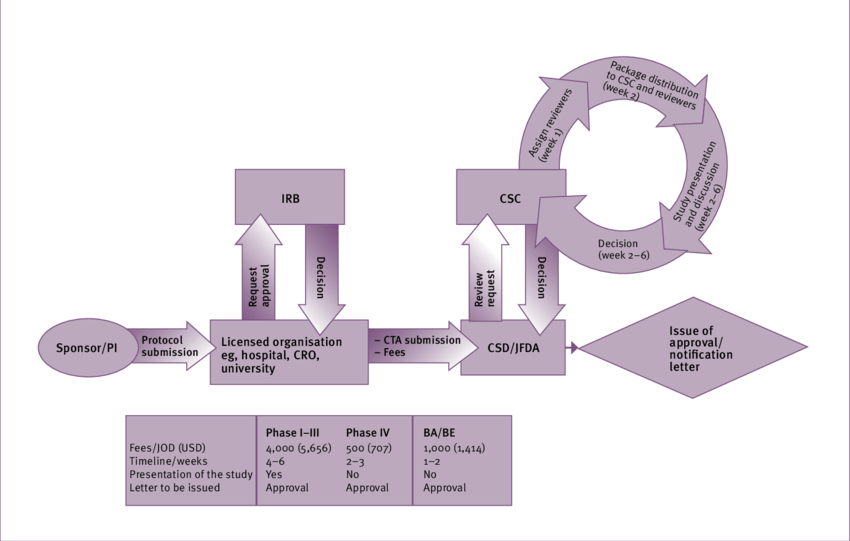
Providing expert support for Clinical Trial Applications (CTA), including protocol review, regulatory submission, and approval facilitation, ensuring compliance with regulatory standards and trial initiation.
Validation and Quantifying

Help clients ensure that pharmaceutical processes, equipment, and systems consistently meet regulatory standards, maintain quality, and operate reliably for safe and compliant production.
Pharmacovigilance Solution
Guiding clients on comprehensive pharmacovigilance solutions, including risk assessment, safety reporting, and regulatory compliance, ensuring patient safety and adherence to global pharmaceutical standards.